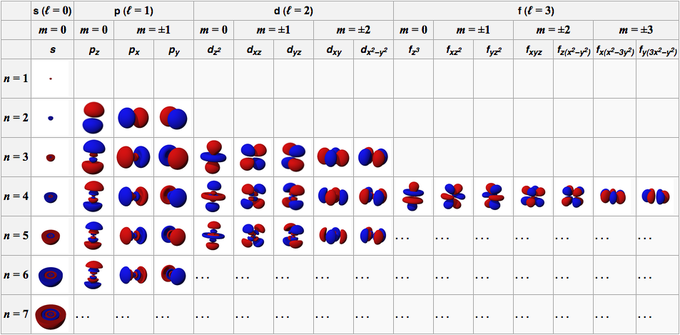
Each type of orbital can hold a specific number of electrons based on its capacity. How do you find electrons in orbitals? The number of electrons in an orbital depends on the type of orbital (s, p, d, f) and the energy level.They can be derived from the electron’s energy level (n), shape (l), orientation (ml), and spin (ms). How do you find the quantum number? Quantum numbers are determined by the electronic structure of an atom.Each orbital can accommodate a maximum of 2 electrons (one with spin up and one with spin down). How many electrons does n = 6, l = 3, ml = -1 have? The quantum numbers n = 6, l = 3, and ml = -1 represent a specific orbital in the sixth energy level.How do you write 4 quantum numbers for an electron? The four quantum numbers are written as (n, l, ml, ms), representing the principal, azimuthal, magnetic, and spin quantum numbers, respectively.What is the quantum number of the 2p electron? The quantum numbers for a 2p electron are n = 2, l = 1, ml can be -1, 0, or 1, and ms can be +1/2 or -1/2.How many electrons are in n = 4? In the fourth energy level (n = 4), there can be a maximum of 32 electrons distributed among different subshells.How many electrons in a quantum number? A quantum number doesn’t directly represent the number of electrons it characterizes different aspects of an electron’s behavior within an atom.The principal quantum number (n), azimuthal quantum number (l), magnetic quantum number (ml), and spin quantum number (ms) collectively provide information about the location, orientation, and spin of electrons within an atom. How do you find electrons from quantum numbers? Quantum numbers describe the properties and energy levels of electrons in an atom.Observations of the interstellar medium reveal atomic hydrogen spectral lines involving n on order of hundreds values up to 766 were detected. In atomic physics, higher n sometimes occur for description of excited states. In chemistry, values n = 1, 2, 3, 4, 5, 6, 7 are used in relation to the electron shell theory, with expected inclusion of n = 8 (and possibly 9) for yet-undiscovered period 8 elements. 
However, the modern theory still requires the principal quantum number. 
With the development of modern quantum mechanics, the simple Bohr model was replaced with a more complex theory of atomic orbitals. The principal quantum number was first created for use in the semiclassical Bohr model of the atom, distinguishing between different energy levels. Description of energy levels based on n alone gradually becomes inadequate for atomic numbers starting from 5 ( boron) and fails completely on potassium ( Z = 19) and afterwards. For multielectron atoms this splitting results in "subshells" parametrized by ℓ. In more complex systems-those having forces other than the nucleus–electron Coulomb force-these levels split. In a simplistic one-electron model described below, the total energy of an electron is a negative inverse quadratic function of the principal quantum number n, leading to degenerate energy levels for each n > 1. Accounting for two states of spin, each n- shell can accommodate up to 2 n 2 electrons. For each value of n there are n accepted ℓ (azimuthal) values ranging from 0 to n − 1 inclusively, hence higher- n electron states are more numerous. For higher n the electron is farther from the nucleus, on average. Its values are natural numbers (from 1) making it a discrete variable.Īpart from the principal quantum number, the other quantum numbers for bound electrons are the azimuthal quantum number ℓ, the magnetic quantum number m l, and the spin quantum number s.Īs n increases, the electron is also at a higher energy and is, therefore, less tightly bound to the nucleus. In quantum mechanics, the principal quantum number (symbolized n) is one of four quantum numbers assigned to each electron in an atom to describe that electron's state. Quantum number assigned to each electron in an atom to describe that electron's state
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
